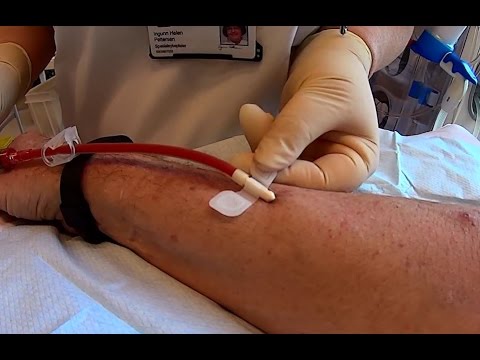
As most Chronic Kidney Disease patients who conduct Dialysis know, the Buttonhole (Constant-Site) Technique is a method which inserts a Dialysis Needle into the same spot each time Dialysis Treatments are conducted. This allows scar tissue to form at the insertion point and creates a tunnel or hole, similar to the hole in a pierced ear. Used for almost 30 years this "Needle Sticking" Technique has been shown to cause fewer complications, decreased pain, and is less time consuming when compared to using different needle insertion sites for each Dialysis Treatment.
Recommended Reading: FDA Says Germ-Killing Soap Recommended For CKD Patients Could Actually Pose Severe Health Risks
The BioHole Dull Anti-Stick Needle received its Food and Drug Administration (FDA) approval in 2010. The company brags that it has a Sterile Scab Remover to assist in preparing the access, and suggests that it is an, "excellent option for someone doing repeated cannulation in the same access site." However, Home Dialyzors United has now asked the FDA to "revoke its approval of the needle" after several patients suggested that the needle is "difficult" and "pose significant risks to the health and safety of patients receiving Dialysis."
Recommended Reading: FDA Warns Dietary Supplements For CKD, Dialysis & Diabetic Patients May Risk Invasive Fungal Disease
Major concerns include: The BioHole Needle being a quarter of an inch (1/4) longer which makes it easier for patients to puncture their own fistulas. Patients and Home Dialyzors United also noted that since the Scab Remover was not packaged separate from the needle, once the needle wrap is opened, the Scab Remover which is similar to a ballpoint pen cap, may be exposed to non-sterile conditions - risking infection in a Dialysis patient's AV Fistula Access Site.
Recommended Reading: Life Saving Dialysis Alert Approved By FDA For Hemodialysis Chronic Kidney Disease Patients
What's more, patients expressed concern with the fact that the BioHole Needle is frosted and they cannot ensure that their Dialysis Line is "Bubble Free." Introducing an air bubble into a patients vein (Air Embolism) could cause air to move through the body and block a small artery; cutting off the blood supply to a particular area of the body. Hence, patients and Patient Care Technicians must remain on alert for air bubbles.
Recommended Reading: FDA Announces Promising Breakthrough New Drug To Fight Common Infection In CKD & Dialysis Patients
Complications were also outlined with the Scab Remover. Home Dialyzors United found that, "The scab picker is not shaped to remove the scab easily and completely for access to the Fistula, and may, in fact, push pieces of the scab back into the Fistula." Needless to say, the Dialysis Community does not seem at all pleased with this new Dialysis Needle Technology which actually appears to be growing in use.
Recommended Reading: FDA Approved Drug Therapy To End CKD Patient Anemia: "Biggest Thing To Hit Dialysis In 25 Years."
The company, Nipro, replied to these criticisms in a direct and pithy statement, "The issues that you have outlined in your letter can be significantly reduced with training provided by the Nipro Clinical team through the Nipro Connect Educational Program." What do you think? Should the FDA revoke its approval of the BioHole Needle? Click here to share your opinion with the over 21,900 other Friends at the KidneyBuzz.com Facebook Fan Page. Also, remember to visit KidneyBuzz.com every day for the Latest Breaking News and Information which teaches those with Chronic Kidney Disease and Diabetes how to better manage and improve their lives.